J j thomson cathode ray experiment4/7/2023 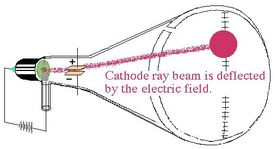
They also discovered that the quantity of voltage, kind of gas, and constituent ratios did not affect the natural, physical, and behavioral features of electrons. The theory helped future physicists in understanding the modern structure of an atom. Thomson derived a conclusion after the experiment that the rays that moved from the cation to the anion were negative. 
The phosphorescent substance was put in such a way that when the rays strike it, little sparks of light appear, allowing the stream of rays to be detected.The cathode rays begin deflecting and repelling from the dipole and moving towards the anode as a result.A dipole is used to detect and measure the beam that was created.A closed-loop circuit propagates electric current.Ionization of partial air inside the terminal due to high voltage causes gas to become the conductor.The equipment is set up so that the terminals have a high voltage and the internal pressure is decreased by eliminating air from the tube.The cathode ray was redirected away from the negatively charged plate and towards the positively charged plate. Thomson used two opposingly charged electric plates around the cathode ray to test the particles' characteristics. Thomson, a scientist, began working with cathode ray tubes in the late 1800s. Cathode rays are not visible, but they were first identified in early vacuum tubes when they impacted the tube's glass wall, energizing the atoms and causing them to emit light-a glow known as fluorescence.In simple words, rays traveling from the negative end to the positive end of an electrode within a vacuum are known as Cathode Rays.A focused beam of electrons is deflected by magnetic or electric fields in cathode ray tubes, in order to produce the image in a traditional television set (CRTs).Ĭathode Rays used to produce images in traditional Television The constituents of cathode rays were the first to be found. When a voltage is given to an evacuated glass tube with two electrodes, electrons emitted from the cathode cause the glass opposite the negative electrode to glow. In vacuum tubes, cathode rays (electron beams or e-beams) are electron streams.When the electron beam hits the phosphor-coated screen, it creates a small, brilliant visible spot on the fluorescent screen. Deflecting coils are of two types: horizontal and vertical.Deflecting coils generate an extremely low-frequency electromagnetic field that allows the electron beam's direction to be adjusted continuously.The electron gun or heater produces a narrow electron beam.CRTs are used in the majority of desktop computer displays.Īs seen in the image, a cathode ray tube is made up of numerous fundamental components: In 1913 Thomson published an influential monograph urging chemists to use the mass spectrograph in their analyses.A cathode ray tube(CRT) is a kind of vacuum tube producing images when an electron beam collides with a phosphorescent surface. 
His nonmathematical atomic theory-unlike early quantum theory-could also be used to account for chemical bonding and molecular structure (see Gilbert Newton Lewis and Irving Langmuir). Of all the physicists associated with determining the structure of the atom, Thomson remained most closely aligned to the chemical community. He was a good lecturer, encouraged his students, and devoted considerable attention to the wider problems of science teaching at university and secondary levels. Even though he was clumsy with his hands, he had a genius for designing apparatus and diagnosing its problems. In 1884 he was named to the prestigious Cavendish Professorship of Experimental Physics at Cambridge, although he had personally done very little experimental work. He was then recommended to Trinity College, Cambridge, where he became a mathematical physicist. Instead young Thomson attended Owens College, Manchester, which had an excellent science faculty. His father intended him to be an engineer, which in those days required an apprenticeship, but his family could not raise the necessary fee. Ironically, Thomson-great scientist and physics mentor-became a physicist by default. From "The Growth of Physical Science," by Sir James Hopwood Jeans (Cambridge: Cambridge University Press, 1948) Early Life and Education
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
